Indianapolis, Indiana – Attorneys for Plaintiff, Blue Sky Networks, LLC of Plano, Texas filed suit in the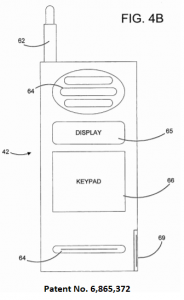 Southern District of Indiana alleging that Defendant, Roche Diabetes Care, Inc., of Indianapolis, Indiana infringed its rights in United States Patent Nos. 6,484,027, (“the ‘027 Patent”), 6,865,372, (“the ‘372 Patent”), 8,265,691, (“the ‘691 Patent”), 8,346,169, (“the ‘169 Patent”), and 8,792,828 (“the ‘828 Patent”). Plaintiff is seeking judgment, damages, and attorneys’ fees.
Southern District of Indiana alleging that Defendant, Roche Diabetes Care, Inc., of Indianapolis, Indiana infringed its rights in United States Patent Nos. 6,484,027, (“the ‘027 Patent”), 6,865,372, (“the ‘372 Patent”), 8,265,691, (“the ‘691 Patent”), 8,346,169, (“the ‘169 Patent”), and 8,792,828 (“the ‘828 Patent”). Plaintiff is seeking judgment, damages, and attorneys’ fees.
According to the complaint, “The Asserted Patents are directed to wireless mobile devices, such as handsets, peripherals, and computing devices, that operate via wireless short-range direct communication with other wireless devices. Such devices may also be enabled for simultaneous operation on a wireless network…and wireless short-range direct communication with other wireless devices.”











